In this radio interview with Dr. Ronald Hoffman, Dr. Thomas Guilliams offers a critique of recent studies on multivitamins, highlighting methodological flaws and bias: http://www.drhoffman.com/podcasts/channel-1/drhoffman-com-2014-01-02-123.mp3
Author Archives: pointadmin
The case is closed: Editorial bias prevents reasonable evaluation of dietary supplements!
Being involved with dietary supplement research for almost 18 years, I have witnessed my share of hype for, and against, the use of dietary supplements. Few, however have attempted such blatant finality to the subject as the recent editorial in the Annals of Internal Medicine– titled “Enough is Enough: Stop wasting money on vitamin and mineral supplements.” The editorial, coupled with the publication of three papers in the same issue, declares in no uncertain terms that “..we believe the case is closed-supplementing the diet of well-nourished adults with (most) mineral or vitamin supplements has no clear benefit and might even be harmful. These vitamins should not be used for chronic disease prevention. Enough is enough.” The publication of the editorial was hyped by many news outlets who quickly found the usual supplement bashers, all too willing to add insult to injury by regurgitating decades-old sound-bites.
Anybody that has spent even a brief amount of time evaluating medical research, especially as it pertains to the use of vitamins and minerals, knows that such a conclusion (“the case is closed”) is as arrogant as it is absurd. In fact, the editorial doesn’t even do justice to the data presented in the three papers published within the same issue- let alone the broader evidence used to support the use of certain vitamins and minerals for the prevention of chronic disease. Let us briefly discuss the 3 papers published in this particular Annals issue- before moving on to the broader context that may expose the real issue behind this editorial. They included [1] the vitamin-only and placebo-only arms of the TACT (Trial to Assess Chelation Therapy), looking for reduced event rates after an initial myocardial infarction, [2] an attempt to see measurable cognitive changes when giving men a multivitamin- part of the Physicians Health Study II, and [3] a systematic review of a select group of studies using various vitamin preparations for primary prevention of cardiovascular disease and cancer- a review prepared for the U.S. preventative Services Task Force.
1. We have previously discussed the initial data from the TACT trial [here]- which showed statistically lower events when patients were given EDTA chelation, compared to placebo.[1] Since this study was a 2×2 factorial trial- the two arms receiving no EDTA chelation (i.v. saline/placebo) given either placebo capsules or high-dose multivitamin capsules were compared in this study.
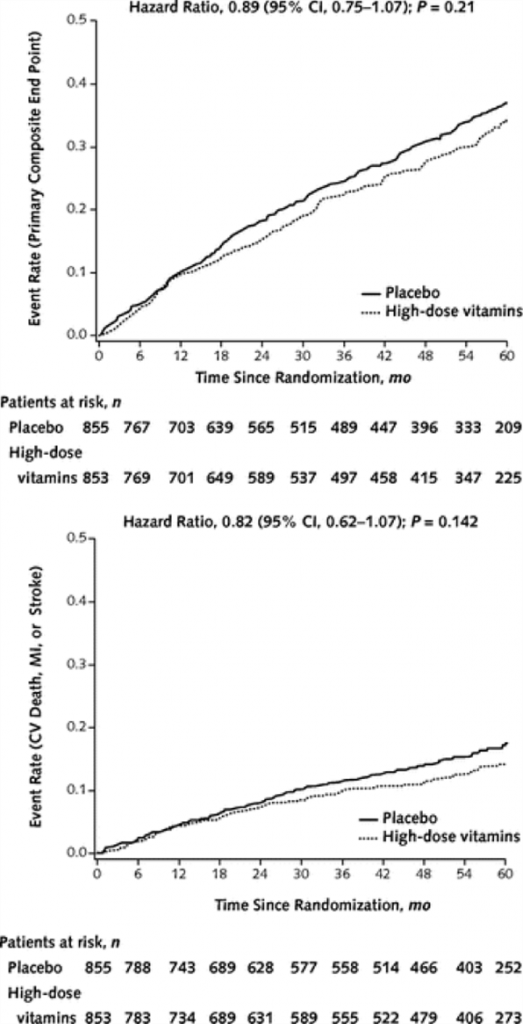
A high-dose vitamin supplement, similar to those sold by a number of physician-only product companies, was used for this study. Patients were at least 50 years old and all had sustained a previous myocardial infarction. The primary end-point was a composite of time to death from any cause, re-infarction, stroke, coronary revascularization or hospitalization for angina. A secondary end-point using just cardiovascular death, re-infarction and stroke was also assessed. According to the authors, the high-dose vitamins showed an 11% relative reduction in the primary end point compared to placebo, but this difference did not reach statistical significance (see the cumulative events recorded over 60 months in the primary (top) and secondary (bottom) outcomes in the adjacent figure). The authors make it clear to us that while the trial does not support the routine use of high-dose oral multivitamin regimen for all patients who have had an MI, the total number of events were smaller than the trial was originally powered to detect and thus -“ the reduced statistical power due to a small difference between groups, as well as the nonadherance to the study regimen, limits the conclusion of nonefficacy.” [emphasis added]. The number of people who stopped their vitamin or placebo therapy was staggeringly high at 46%.
Let us now review a few more things that might be of interest to you- but left out of the editorial. First is the fact that both the vitamin group and the placebo group were consuming a high number of pharmaceutical agents (as one might expect of a post-infarct cohort). Subjects were on aspirin (>82% of subjects), beta-blockers (70%), statins (70%), ACEi/ARB (60%), Clopidogrel (25%), and oral hypoglycemic (>20%). On top of this- nearly half the patients were taking other multivitamin supplements! So, in essence, this study was looking for a statistical difference between one group of subjects taking high-dose vitamins (46% of whom discontinued taking them) and another group of subjects (half of whom were consuming another multivitamin of unknown ingredients)- while both groups consumed high amount of pharmaceuticals which are known to reduce both the primary and secondary end-points measured.
Furthermore, there was one important cohort that realized a statistically significant reduction in the primary end-point when given the multivitamins; those individuals not on a statin drug (38% reduction in events p=0.012). So when we remove the effect of statins on these subjects- we see a strong benefit of the supplemental nutrients (these authors go on to tell to ignore these results, though they are both clinically and statistically significant, until more studies can be done). A very strong trend also existed in patients enrolled less than 5 years since their MI (p=0.046), suggesting that these nutrients are less effective the longer an individual has been pharmacologically-treated since their MI. Finally- while this study used a supplement containing much higher doses than nearly any other multivitamin trial to date- it is important to note that, unlike the widely repeated concerns of risk, this trial reported no difference in severe adverse events or incident cases of cancer.
2. The second paper was a sub-study of the Physicians’ Health Study II (PHS2). Previous analysis of this data already showed that this multivitamin therapy statistically reduced the risk for cancer and cataracts.[2] In this analysis, cognitive function was measured using the Telephone Interview for Cognitive Status (TICS). Although the PHS2 involved 4 arms, the data presented here was between two group given either a multivitamin (Centrum Silver) or placebo. Subjects over 65 were recruited from within the PHS2 for this sub-study. They found, after 4 such telephone assessments over 12 years, that there was no statistical difference between the two groups in the mean level of cognition. They conclude that “in male physicians aged 65 years or older, long-term use of a daily [Centrum Silver] did not provide cognitive benefits.”
The limitations of this study are many. First, because this was a sub-study of the PHS2, the first (baseline) cognitive test began an average of 2.5 years after patients were randomized to their multivitamin or placebo. This means that the baseline could have been already influenced by years of the therapy. Even though their baseline data showed no statistical, between group differences, this fact alone would likely prevent anyone from drawing firm conclusions from this data. On top of this, subjects in the PHS2 were only prevented from taking other multivitamins if those products contained more than the USRDA of vitamin E, vitamin C, B-carotene or vitamin A. Which means they could be consuming high levels of B-vitamins- known to lower homocysteine (a metabolite associated in some studies with cognitive risk), or any of a number of other supplements known to affect cognition (Ginkgo biloba, phosphotidylserine, omega-3 fatty acids, vinpocetine, etc.) without the knowledge of the researchers (up to 1/3 of subjects were taking other multivitamins in the PHS2). These gross oversights are due to the simple fact that the PHS2 was obviously not designed to answer whether daily multivitamin use affects cognitive function in healthy older physicians in the first place.
Since few observational studies have examined the relationship between multivitamin use and cognition; and since the PHS2 was also not originally designed to ask this question; these data do not allow any broad conclusions about the benefits of all multivitamins (and doses) on potential cognitive benefits. While we don’t typically recommend products like Centrum Silver- it should have been obvious to these researchers that this product was not specifically designed to modulate cognitive function in healthy 65 year old male physicians; nor were there previous trials to suggest such an outcome. It is curious that a study (PHS2) not designed for this primary end-point, coupled with an intervention (Centrum Silver- one/day) not designed for this primary end-point can be evidence for anything- let alone for an argument that the “case is closed.”
3. The last of the three published articles is a systematic review of the benefits and harms of vitamins and mineral supplements in community dwelling, nutrient-sufficient adults for the primary prevention of either cardiovascular disease or cancer. After weeding through thousands of potential articles, these reviewers selected 103 articles (representing only 26 studies) that fit their study selection criteria. As one would expect, these trials varied considerably in study design, recruitment criteria and primary end-points; and most importantly- differed dramatically in the multivitamins or mineral products used in each study. Not surprisingly, they “found no consistent evidence that the included supplements affected CVD, cancer, or all-cause mortality in healthy individuals without known nutritional deficiencies.”
Rather than attempt to parse the nuances of each selected study, a broader critique will be sufficient for this review. Blinded by their desire to debunk the use of “vitamins” and “minerals”- these reviewers ignore the fact that each nutrient has a completely different mechanism of action, therapeutic dose potential and historical data. Comparing studies where subjects consumed the hormone-like cholecalciferol, with studies using the water-soluble antioxidant ascorbic acid, merely because both are classified as “vitamins,” is absurd and unscientific. On top of that, they excluded from their analysis any studies that used doses higher than the upper tolerable limit set by the U.S. Food and Nutrition Board. This would exclude products with more than 4000IU vitamin D, 35 mg of niacin, 1 mg of folate, or 350 mg of magnesium- often exceeded in products known for their therapeutic benefit. Furthermore, since they excluded studies where subjects were nutrient deficient- this virtually eliminates the application of this data to “average” American- many of whom are deficient in more than one vital nutrient; which begs the question of this review’s intended application.
The authors do admit that this review study design is used “primarily to evaluate drug therapy. The design might not be ideally suited to evaluate nutrients.” They also acknowledge that since subjects in the placebo arm of each of these studies are healthy and not known to have any nutrient deficiencies- they are, in fact, comparing subjects with “adequate” nutrient intake (placebo) versus those with higher than adequate nutrient intake (supplemented treatment). In many cases, the “placebo” group is not even prevented from taking other supplements; or are consuming higher amounts of nutrients than the researchers originally anticipated (they note, for instance, that Women in the WHI control group had twice the average calcium intake than the study design anticipated).
In the end, this highly selected review of widely divergent low-dose studies (only a few which reflect “real-world” supplementation) adds little to the evaluation of the use of appropriately dosed nutrient supplements for reducing the risk of (i.e. preventing) chronic disease.
A final perspective: I find it ironic that while the FDA demands that manufacturers of dietary supplements constantly reassure their customers that “these products are not intended to cure, treat or prevent any disease”- that this same statement (although almost certainly untrue) is somehow proof of no health benefit. What this statement, and these types of trials prove- is that, alas- nutrients are not drugs! Furthermore, the studies designed to “prove” drug efficacy are inadequate and inappropriate to evaluate the benefits of nutrients. What started out as “evidence-based medicine” has now morphed into “medicine-based evidence”- where drug companies set the rules and FDA gladly enforces them. And even though the $300 billion pharmaceutical industry is 10-times larger than the supplement industry (only ~40% of which is vitamins)- we are advised to “stop wasting our money” only on the latter. Thankfully, the American public knows better- even if they don’t have access to all the nuances uncovered here. For them, the case is also closed: and they are not going to wait for the paid researchers to finally figure it out.
- Lamas GA et al. Effect of disodium EDTA chelation regimen on cardiovascular events in patients with previous myocardial infarction: the TACT randomized trial. JAMA. 2013 Mar 27;309(12):1241-50.
- Gaziano JM, Sesso HD et al. Multivitamins in the prevention of cancer in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2012 Nov 14;308(18):1871-80.
- Rautiainen S, Wang L, Gaziano JM, Sesso HD. Who uses multivitamins? A cross-sectional study in the Physicians’ Health Study. Eur J Nutr. 2013 Oct 30
If you want to know when Dr. Guilliams posts future blogs or when a new whitepaper is available, “Like” us on our Facebook page. To get Dr. Guilliams’ book The Original Prescription– you can purchase directly from the Point Institute or purchase on Amazon.
New Technical Report on choosing the best Omega-3 product for therapeutic use
Dr. Guilliams’ latest Technical Report reviews the evidence supporting fish oil as the standard marine-derived omega-3 fatty acid, and discusses the various product types, their bioavailability, ethyl esters vs. triglycerides, pertinent quality control issues and gives take home recommendations. Since the wide range of therapeutic uses and forms, as well as sources and delivery forms of marine-derived omega-3 fatty acids is a source of confusion for patients and clinicians alike, this Point Institute paper reviews the evidence and explains the data supporting the use of fish oil, which has become the known standard in the world of marine derived Omega 3 products. The paper discusses the various types of marine derived Omega 3 products and the considerations which should be made when selecting one. Other topics include common concerns, such as allergies to fish oil, and the difference between Krill vs. Fish oil sources. The paper also discusses the research on the bioavailability of various structures and forms such as ethyl esters vs. triglyceride, as well as the differential uses of DHA and EPA, what defines pharmaceutical grade products, quality control issues, and ends with take-home recommendations for product selection.
.
Why water should be your primary beverage
With the recent discussion of water intake and health- here is an excerpt from The Original Prescription that discusses the principle of using water as our main source of hydration and a little bit about the origins of the 8 glasses per day “rule.”
This is just one of over 50 Lifestyle principles discussed in the Original Prescription.
Principle #6: Water should be your primary beverage; drink enough, and try to limit the number of liquid calories you consume.
Water is one of the quintessential nutrients of life and yet is often one of the most commonly neglected. Despite its great importance for nearly every bodily function, many of us are still at a loss when it comes to knowing how much we’re supposed to drink each day and which other beverages count toward our daily needs. All this confusion about water needs comes as no surprise considering that the first official recommendation for adequate intake (AI) of water was only established in 2004 by the Institute of Medicine, coinciding with an increase in popular awareness about water needs for preventing conditions such as cancer, heart disease, and weight gain (27). Prior to that, the RDA concluded that it was impossible to set a recommendation for water needs, and the National Research Council used a general rule of thumb of 1 ml/ kcal (that would be two liters of water per 2000 calories consumed, if you’re getting out your calculator) (28). Today’s recommendations for daily water requirements, however, are based on national averages from the NHANES III data, although individual needs vary greatly. It’s estimated that most of us need to get 80% of our daily hydration through beverages, mostly water, while about 20% of our hydration comes from the food we eat (less if you happen to avoid fresh fruits and vegetables) (28).
Throughout history, humans have relied on water as their primary source of hydration, struggling to ensure both an abundant and a clear source was available for their survival. Unfortunately, this struggle still exists in many locations around the world today. In the West, water abundance (at least for drinking) is rarely in jeopardy, and yet many choose other options. Countless individuals have replaced water with soft drinks to quench their thirst, and, as a result, sweetened beverages have become one of the major sources of calories in the American diet. Consumption of high fructose corn syrup, the major sweetener in commercial soft drinks, increased over 1000% between 1970 and 1990, and today, half of all Americans consume soft drinks every day. In fact, these beverages now constitute the leading source of added sugar in the average diet (29,30). To make matters worse, the calories provided by soft drinks often fail to satisfy hunger the way solid food does, nor do they quench our thirst in the way water can, making sugary beverages a key player in the obesity epidemic (30–33). If you want to maintain those good signals that your body is waiting for, limit the number of calories you consume through drinks. So how much water should you drink per day? Well, that depends. How much you weigh (roughly 60% of that is water), how much water you lost recently to perspiration, and relative humidity will all affect the ultimate answer. The Institute of Medicine says that the adequate intake of total water per day is 3.7 liters for men and 2.7 liters for women.† The “8 by 8 rule” (8 glasses of 8 oz. each) equates to about 2 liters. Many rely on their thirst to tell them when to drink, and while it is true that most people’s thirst and hunger mechanisms can help manage their net water balance, in many people the thirst mechanism is blunted and mild dehydration can set in well before their body tells them to drink. Others try to rely on the color and darkness of their urine as a gauge of hydration, but this is not always a reliable indicator of the need for water (35)
So what’s the bottom line when it comes to hydration and water?
- If you aren’t already doing it, drink mostly water to keep yourself hydrated.
- Make sure your water source is clean and free of contaminants.
- Tea and coffee are fine for most people (in moderation); if the caffeine causes you to urinate frequently, consider offsetting this loss with additional water.
- Alcohol is dehydrating—plain and simple.
- Remember to include water-based soups and stews, herbal teas, and low-sugar fruit juices.
- Drink more water when you are physically active and when the weather turns hotter.
† The AIs provided are for total water in temperate climates. All sources (according to IOM) can contribute to total water needs: beverages (including tea, coffee, juices, sodas, and drinking water) and moisture found in foods. Moisture in food accounts for about 20% of total water intake.
Get your copy of The Original Prescription on our Website or on Amazon.
Fish Oil and Prostate Cancer Risk- Is There a Link?
Several widely publicized clinical trials in the past weeks and months purport that the use of omega-3 fatty acids, especially those from supplemental fish oil, has no therapeutic value or are even potentially harmful to consume. This short communication [pdf of this article] is written to help put these reports within the context of the studies’ own limitations and also the broader fish oil research that has gone virtually unreported during the same time.
Let’s begin with the most recent study purporting to link fish oil with prostate cancer risk. The report was published online in the J. of the National Cancer Institute (Online Abstract) and soon afterward, headlines such as “Men who take fish oil omega-3 supplements at 71% higher risk of prostate cancer: study”[1] began floating around the internet, print and national television. One would assume by such headlines that this study was specifically designed to look at prostate cancer risk in men consuming fish oil supplements- it was not. In fact, this study didn’t even look at fish oil (or even dietary fish) consumption in these subjects!
Instead, this report was a secondary analysis of data collected from the previously concluded SELECT (Selenium and Vitamin E Cancer Prevention Trial) trial, which recruited men over 50 with no history of prostate cancer who were then randomized to receive vitamin E, selenium, a combination of vitamin E plus selenium, or placebo and followed for prostate cancer incidence (NCI’s SELECT Website). The data from this report is considered a case-cohort design, attempting to find an association between plasma phospholipid fatty acid levels in case subjects (those diagnosed with prostate cancer during the length of the trial) and compare those with study cohorts (similar subjects within the trial not diagnosed with prostate cancer during the length of the trial). It is important to note that the original vitamin E/selenium study was not designed to detect the association between plasma phospholipids and cancer risk and patients were not asked about their fish or fish oil supplement use at either the start or the length of the trial.
Nevertheless, they report that higher levels of 3 specific omega-3 fatty acids (EPA,DHA,DPA- combined) as a percent of plasma phospholipid fatty acids- were associated with a higher incidence of prostate cancer during the SELECT trial. While the authors are much more cautious in their direct indictment of omega-3 supplementation within their publication (since they have no data related to supplementation), one of the authors said in a press release “We’ve shown once again that use of nutritional supplements may be harmful” Here are a few reasons these conclusions and statements are unwarranted and misrepresent the data.
- The fatty acid levels reported here represent only a single blood draw taken at the start (baseline) of each participant’s entry into the study, often years before a prostate cancer diagnosis was assigned to the subject. Since plasma phospholipid fatty acid content fluctuates with dietary intake on a day to day basis, a single time point may only reflect dietary habits within the previous week prior to the blood draw and may have no correlation to long-term omega-3 intake or blood levels.
- While the omega-3 fatty acid differences between groups were statistically different, they were not clinically significant. That is, the omega-3 levels reported would be considered “average” in all subject groups and the largest difference in the levels reported between groups could have been achieved with very low omega-3 consumption in a few weeks’ time.(see endnote #2)[2]
- The authors admit that because of the high cost of phospholipid testing, that only case subjects diagnosed through 2007 (and their cohorts) were originally to be tested. But since “new finding” of associations between fatty acids and prostate cancer came to light- more subjects with high-grade cancer were analyzed in the 8th and 9th year of the trial. This highly unusual change in data set would have been disallowed in most other peer-review settings. The original data set was not published.
- In almost all cases of associative data, a number of variables are used to adjust the data. Typically these adjustments include most variables that might influence risk. While these data were adjusted for education, diabetes, family history of prostate cancer and the SELECT intervention assignment; these data were not adjusted for the most striking variables that affect risk in this population- age, race, BMI and PSA levels- information which may have nullified these statistical associations. How these reviewers ignored this most obvious adjustment and permitted the data to be reported without these adjustments is baffling.
Omega-3 associated with lower prostate cancer, breast cancer, CHD and total mortality.
Beyond the specific conclusion of this study is the broader epidemiological and scientific question of plausibility. The authors readily admit that there is no plausible scientific explanation for how long-chain fatty acids like EPA and DHA could actually cause prostate cancer. There is also the inconvenient fact that several people groups which consume high levels of omega-3 fatty acids and have plasma phospholipids much higher than the participants in this study have extremely low incidence of prostate cancer (i.e. Japan). More importantly, studies that specifically look at fish and fish oil consumption show a dramatic decrease in prostate cancer risk in older men, seeing a slightly higher risk associated only with salted and smoked fish intake. [3] Other large meta-analysis have shown that even when overall prostate cancer incidence may be unaffected by omega-3 intake, prostate cancer mortality is dramatically lower in individuals with higher intake of marine omega-3 fatty acids.[4]
In fact, in a highly under-reported study published this year in the Annals of Internal Medicine, plasma phospholipid omega-3 fatty acids (much like the SELECT data above) were associated with lower total mortality- especially related to CHD deaths.[5] However, in this study the plasma phospholipid fatty acid difference between the highest and lowest groups were highly clinically relevant (200-300% difference), as compared to the clinically irrelevant differences in the SELECT trial data (6% difference-see endnote #2).
Lost in all this has been another significant report published in the British Medical Journal which associates the intake of fish and marine omega-3 fatty acids with a reduced risk for breast cancer.[6] This meta-analysis of 21 independent prospective cohort studies showed an overall 14% reduction in the relative risk for breast cancer related to marine omega-3 fatty acids. This risk reduction was associated with both the consumption of fish and fish oil, as well as tissue biomarker analysis (i.e. plasma phospholipids). They even suggested a “dose-response” relationship which suggested that the risk of breast cancer was reduced by 5% for each 100mg/day of marine omega-3 consumed.
Summary:
When the data from the subcohort of the SELECT trial is analyzed and placed alongside the growing epidemiological, interventional and mechanistic data (see below) – the purported relationship between consuming omega-3 fatty acids (in the diet or through dietary supplements) with an increased risk of prostate cancer cannot be supported. Furthermore, since this study did nothing to ascertain the consumption of fish or omega-3-containing supplements in these subjects, it makes the sensationalized media reports about this study even more disturbing. In addition, the fact that the association data was not adjusted for the most obvious factors (such as PSA levels, race, BMI and age) leaves us unsure that there is any association at all; and leaves many others to suppose that strong bias may be at play here. We find it quite telling that one of the key authors of the trial is quoted in the DailyMail as saying ‘There is not really a single example of where taking a supplement lowers chronic disease risk.’ [7] On the contrary, we believe the current overall scientific evidence suggests that consumption of omega-3 fatty acids from fish oil supplements is not only safe at a wide-range of doses, but has proven efficacy in reducing risk for a wide-range of chronic conditions.
Recent animal or basic research on omega-3 fatty acids and prostate cancer
- Mechanisms of omega-3 polyunsaturated Fatty acids in prostate cancer prevention. Biomed Res Int. 2013;2013:824563.
- A low dietary ratio of omega-6 to omega-3 Fatty acids may delay progression of prostate cancer. Nutr Cancer. 2013;65(4):556-62
- 15-lipoxygenase metabolites of docosahexaenoic acid inhibit prostate cancer cell proliferation and survival. PLoS One. 2012;7(9):e45480.
- 22 : 6n-3 DHA inhibits differentiation of prostate fibroblasts into myofibroblasts and tumorigenesis. Br J Nutr. 2012 Dec 28;108(12):2129-37
- Effect of dietary polyunsaturated fatty acids on castration-resistant Pten-null prostate cancer. Carcinogenesis. 2012 Feb;33(2):404-12.
- Consumption of high ω-3 fatty acid diet suppressed prostate tumorigenesis in C3(1) Tag mice. Carcinogenesis. 2012 Jan;33(1):140-8
- Docosahexaenoic acid selectively induces human prostate cancer cell sensitivity to oxidative stress through modulation of NF-κB. Prostate. 2011 Sep 15;71(13):1420-8.
[1] http://www.nydailynews.com/life-style/health/evidence-prostate-cancer-omega-3-link-article-1.1395853
[2] For instance, the greatest difference between DHA levels in these subjects was reported as 0.18% (2.91% in the no cancer group and 3.09% in the high-grade cancer group, difference P=0.009). For comparison, other studies have shown that fish oil intake equivalent to a single serving of fish per week can raise DHA levels 0.63%, and do so in about 12 days. (AJCN 2012; 96:748). As Duffy MacKay, VP of Science & Regulatory Affairs at the Council for Responsible Nutrition said about the most recent study, these difference in omega-3 levels “literally could have occurred if somebody ate a fish sandwich on their way to get their blood drawn”
[3] Consumption of Fish Products across the Lifespan and Prostate Cancer Risk PLoS One. 2013; 8(4): e59799).
[4] Fish consumption and prostate cancer risk: a review and meta-analysis. Am J Clin Nutr. 2010 Nov;92(5):1223-33
[5] Plasma phospholipid long-chain ω-3 fatty acids and total and cause-specific mortality in older adults: a cohort study. Ann Intern Med. 2013 Apr 2;158(7):515-25.
[6] Intake of fish and marine n-3 polyunsaturated fatty acids and risk of breast cancer: meta-analysis of data from 21 independent prospective cohort studies. BMJ. 2013 Jun 27;346:f3706
Dr. Guilliams featured in Stevens Point Journal
In the Stevens Point Journal interview, Dr. Thomas Guilliams is interviewed about the Seven Pillars of good health, as discussed in The Original Prescription book (video included): http://www.stevenspointjournal.com/apps/pbcs.dll/article?AID=2013306260186
CoQ10- Dramatic benefit for heart failure (and update)
Coenzyme Q-10 has been used as a dietary supplement for a variety of uses over the past few decades. Among its many therapeutic benefits, are those related to heart failure symptoms and mortality. This past week the results of a large clinical trial were presented at a congress of the European Society of Cardiology Heart Failure Association. That study (Q-SYMBIO) showed that 300mg of daily CoQ-10 supplementation for 2 years had a highly significant effect on major adverse cardiovascular events (50% reduction) and all-cause mortality (>45% reduction). Here is a news reports about this study.
Since the full paper has not been published, it is difficult to assess the nuances of the data- something that has invited many to discount the potential benefits of these study results. The cautious, but optimistic conclusion of the study authors is that “CoQ10 should be considered as a part of the maintenance therapy of patients with chronic heart failure.”
*- Remember- Statin therapy reduced serum CoQ-10
Here are some more recent studies using CoQ-10 you might be interested in-
- Effect of coenzyme Q₁₀ supplementation on heart failure: a meta-analysis. Am J Clin Nutr. 2013 Feb;97(2):268-75.
- The effect of coenzyme Q10 in statin myopathy. Neuro Endocrinol Lett. 2012;33 Suppl 2:98-101.
- Impact of coenzyme Q-10 on parameters of cardiorespiratory fitness and muscle performance in older athletes taking statins. Phys Sportsmed. 2012 Nov;40(4):88-95.
- Can Coenzyme Q10 Improve Clinical and Molecular Parameters in Fibromyalgia? Antioxid Redox Signal. 2013 Apr 6.
- Coenzyme Q10 deficiency in mitochondrial DNA depletion syndromes. Mitochondrion. 2013 Jul;13(4):337-41.
- Coenzyme Q10 Supplementation Reduces Oxidative Stress and Increases Antioxidant Enzyme Activity in Patients with Relapsing-Remitting Multiple Sclerosis. Int J Neurosci. 2013 May 10.
- Coenzyme q10 regulates osteoclast and osteoblast differentiation. J Food Sci. 2013 May;78(5):H785-891.
Berberine – A Nutraceutical for Glucose and Lipid control: An Update
I was recently reminded of how impressive berberine can be after recently hearing a clinician’s story. The particular patient had Type 2 diabetes and this clinician had been treating him for 15 years. Consistently his glycated hemoglobin was above 9% (aim: to be below 7%). Historically, the patient had been taking 2 grams of Metformin, then recently these meds were lowered to 1 g and berberine was added. Within one month, his glycated hemoglobin was 6.7%. Obviously, this is a single case and other factors may have been involved- lthough the clinician was convinced berberine was a major factor in these results.
We have been following the growing research related to use of the alkaloid compound berberine for almost a decade now and wanted to give a short update for those interested in using this dietary supplement as part of their healthcare protocol for managing glucose, lipids or related cardiovascular outcomes.
Berberine for glucose management in Type 2 Diabetes
Most of the excitement related to the use of berberine for diabetic patients comes from a paper published in early 2010 (Metabolism 59(20)) where a group of Type 2 diabetic patients were given either metformin 1.5 g/d (n=26), rosiglitazone 4 mg/d (n=21) or berberine HCl 1g/day (n=-50) and followed for 2 months. The remarkable results showed drops in fasting blood glucose and glycated hemoglobin for berberine which matched both metformin and rosiglitazone- while only the berberine group saw a statistical drop in TG levels. Furthermore, they followed this study by testing 1g/day of berberine HCl in type 2 diabetic patients who had either hepatitis B or C to detect whether these same outcomes could be confirmed in these patients and what affect berberine may have on liver enzymes in these patients. They found similar benefits in both blood glucose and TG levels as in the non-hepatitis patients, but also a statistical improvement in both AST and ALT enzyme levels.
One of the critiques of this (and most) berberine studies is that they have nearly all been performed in China- on Chinese subjects and most of the published trials are in Chinese- making the data difficult to assess and translate (both linguistically and clinically) into practice here in the US. A recent meta-analysis of these Chinese studies has recently been published (in English) and is available for download online (Evidence-Based Complementary and Alternative Medicine Volume 2012). While showing a consistent positive benefit for berberine in a wide-range of Chinese subjects (14 trials, 1068 subjects), the authors also conclude that many of these trials were poorly conducted and the need for a large, well-controlled and randomized clinical trial is critical. Nonetheless, they found berberine generally safe- listing GI discomfort and constipation (berberine is traditionally used to treat diarrhea in China) as side-effects to treatment in a small number of patients.
Berberine for lipid-altering effects or related cardiovascular outcomes
Years before the glucose-related outcomes were being published, researchers in China had described lipid-altering effects using berberine. Some of these trials are now being performed outside of China in Caucasian subjects. Most recently was a study done at the University of Pavia (Italy) in 144 subjects with low cardiovascular risk (Expert Opin Biol Ther 2013). After a 6-month run-in period of diet and physical activity, patients were randomly given either placebo or berberine (500 mg- twice per day) for 3 months. Patients were taken off their experimental therapy for 2 months (wash-out), and then placed back on berberine or placebo for an additional 3 months. Not only was berberine deemed safe in these individuals, but subjects consuming berberine had reduced total cholesterol, LDL-cholesterol and TG, and increased HDL-cholesterol. All of these benefits were diminished during the washout period but returned once back on berberine.
A small pilot study, done also in Caucasian subjects, was performed here in the US (U. of South Dakota and South Dakota State University) which explored the lipid-lowering effect of berberine (Phytomedicine 2012). Sixteen obese subjects were given berberine (500 mg- 3 times per day) for 12 weeks. As a pilot study, there was no control group. After 12 weeks they saw a modest loss in body weight (avg. 5 lbs/subject) and a significant reduction in total cholesterol (-12.2%) and triglyceride levels (-23%). 2 subjects stopped the protocol due to GI complaints- which may have been due to the higher dose (1.5 grams) over other studies using only 1 gram.
A review and meta-analysis of the Chinese studies looking at the lipid altering effects of berberine is also online (Planta Medica Abstract-2013)
Other Recent Human Studies using berberine
- Berberine ameliorates inflammation in patients with acute coronary syndrome following percutaneous coronary intervention. Clin Exp Pharmacol Physiol. 2012 May;39(5):406-11
- A clinical study on the short-term effect of berberine in comparison to metformin on the metabolic characteristics of women with polycystic ovary syndrome. Eur J Endocrinol. 2012 Jan;166(1):99-105. [Free Online]
Potential for berberine/drug interactions
“CONCLUSIONS: Repeated administration of berberine (300 mg, t.i.d., p.o.) decreased CYP2D6, 2C9, and CYP3A4 activities. Drug-drug interactions should be considered when berberine is administered.”
Animal or Mechanism studies of interest
One of the most impressive aspects of the recent berberine studies is the vast number of animal, tissue and mechanistic studies which have been emerging in the past few years. Listed below is just a few recent studies related to metabolic pathways and outcomes. I will only highlight one particular study because it is a fascinating relationship between a nutraceutical agent, the gut microbiome and human physiology.
One of the main historical uses for berberine (Coptis chinensis and its extracts) within Traditional Chinese Medicine is for intestinal infections and diarrhea. This has led some to wonder if consuming berberine regularly for cardiometabolic outcomes might promote an imbalance within the gut bacteria (microflora). As it turns out, the answer might be yes, but in a very helpful way. In a study published in PLoS ONE (Full Article Free online), researchers show that the prevention of obesity and insulin resistance which occurs in rats fed a high-fat diet are partially mediated by changes to the gut microflora. This mechanism adds to the long list of known metabolic influences that berberine has on metabolic pathways and links to the growing evidence we now have of the relationship between obesity and gut microflora in humans. While the paper is quite technical, it gives some interesting background information and highlights the type of research that will continue to be done to decipher the benefits of berberine and other nutraceutical agents. It also shows us that some agents have activities which don’t even require absorption to result in a clinical outcome.
Here are more studies on berberine that might interest you:
- Berberine chloride improved synaptic plasticity in STZ induced diabetic rats. Metab Brain Dis. 2013 May
- Berberine Attenuates Cigarette Smoke-Induced Acute Lung Inflammation. Inflammation. 2013 Apr 21.
- Berberine inhibits the growth of human colorectal adenocarcinoma in vitro and in vivo. J Nat Med. 2013 Apr 21
- Effect of Berberine on PPAR α /NO Activation in High Glucose- and Insulin-Induced Cardiomyocyte Hypertrophy. Evid Based Complement Alternat Med. 2013;2013:285489.
- Berberine Ameliorates Chronic Kidney Injury Caused by Atherosclerotic Renovascular Disease through the Suppression of NFκB Signaling Pathway in Rats. PLoS One. 2013;8(3):e59794
- Berberine‑attenuated monocyte adhesion to endothelial cells induced by oxidized low‑density lipoprotein via inhibition of adhesion molecule expression. Mol Med Rep. 2013 Feb;7(2):461-5
New video gives key tips on diet and nutrition
Watch Roni Enten give tips from The Original Prescription book on the power of diet and nutrition, the first of the 7 spheres of Lifestyle Synergy, to drive health (click here to watch video!).
Strontium incorporation into bone using Strontium Citrate
Many of you are aware that strontium, a mineral closely related to calcium, has been used in humans in several forms as a bone-building agent. In Europe, the compound strontium ranelate (a salt that uses ranelic acid) is available as a drug (Protos/Protelos). In the US, FDA has not approved strontium ranelate as a drug, but various forms of strontium are available as dietary supplements (most commonly strontium citrate). Our goal here is not to discuss the widely published efficacy of strontium ranelate, but simply to update you on some interesting and recent studies concerning the related compound strontium citrate; in particular, two studies which address the ability of strontium citrate to deliver similar levels of strontium to bones (I have included links to a number of very remarkable recent studies about strontium ranelate at the end for you to follow if interested).
The first is an animal study (in rats) that is similar to many of the studies which first showed (and are still showing) the various ways in which strontium modifies bone structure and prevents bone fractures. What makes this study interesting for our discussion is that it was a head- to-head study between the citrate and ranelate versions of strontium. Rats were given an equivalent amount of strontium (either as citrate or ranelate) or placebo starting at 19 weeks and followed for another 8 weeks. Researchers could measure the amount of strontium incorporated into the bones of the rats using a custom X-Ray fluorescence spectrometer. Post-mortem analysis of several bones was also performed using the same techniques.
What they found was very simple and straight-forward, strontium provided from citrate salts incorporated into bones just as well (or maybe even better) than the strontium provided from ranelate. As it turns out, they had to re-adjust the data because the strontium content of the strontium citrate was higher than they originally figured- and when they made the adjustment, the two were virtually identical (see figure below). Even though it was predicted (by us and others) that the citrate form is essentially equivalent in its ability to deliver strontium to bone tissue- this study goes a long way to confirm this fact, albeit in an animal model.
The second study is actually a very unique case-study of an osteoporotic women who decided to take strontium citrate and allow researchers at McMaster’s University to follow her bone strontium levels for nearly 3 years (this was an earlier report from the some of the same researchers who performed the rat study above). The subject was 68 years of age and had no history of supplementing strontium in any form prior to the study. She began supplementing with strontium citrate pills which provided 340 mg of elemental strontium (as citrate) per pill; taking 2 per day for a total of 680 mg of strontium per day. Using X-Ray fluorescence, researchers measured the changes in strontium levels in her finger and ankle bones. Not only did her bone strontium levels statistically increase after just 5 days, but it continued to increase until it reached a plateau after 1 year (at least in the finger). However, in the ankle, strontium was continuing to accumulate even after 2.5 years of strontium intake. In both cases, the majority of the increase occurs in the first 3-6 months.
The constraints of such a case-report being used for a wide conclusion are obvious, although there has never been a reason to believe that strontium as a citrate salt would perform in a manner different than strontium as a ranelate salt in this, or any, subject. Even so, we take this limited data for what it is worth. Beyond that, the X-ray fluorescence technique described in this paper could open up more opportunities to study the dynamics of strontium accumulation in bone, as this diagnostic technique results in much less radiation than a traditional DEXA scan. [Email communication with the author of the study does confirm they have repeated this analysis in many more subjects and this data will be published in the near future].
Of course, we would like to see more data generated, perhaps a clinical trial similar to those done with strontium ranelate performed using strontium citrate. Finding someone to fund such a trial while no one holds a patent for strontium citrate (or even an exclusive) is daunting- since such a trial would need to have a large number of subjects and be at least 2-3 years long. I do know of a researcher who is willing to do the study if the money is available…any takers?
A full whitepaper on the role of strontium as a bone-building agent will be available in the Whitepapers section soon. Also see The Standard Vol. 6 No. 2 for a discussion of other related nutrients that can be used to support osteoporosis.
- Strontium ranelate–a promising therapeutic principle in osteoporosis. J Trace Elem Med Biol. 2012 Jun;26(2-3):153-6
- A large prospective European cohort study of patients treated with strontium ranelate and followed up over 3 years. Rheumatol Int. 2013 Mar 2
- Cost-effectiveness of strontium ranelate in the treatment of male osteoporosis. Osteoporos Int. 2013 Feb 1. [Epub ahead of print]
- Healing of subtrochanteric atypical fractures after strontium ranelate treatment. Clin Cases Miner Bone Metab. 2012 Sep;9(3):166-9
- Efficacy and safety of strontium ranelate in the treatment of knee osteoarthritis: results of a double-blind, randomised placebo-controlled trial. Ann Rheum Dis. 2013 Feb;72(2):179-86.